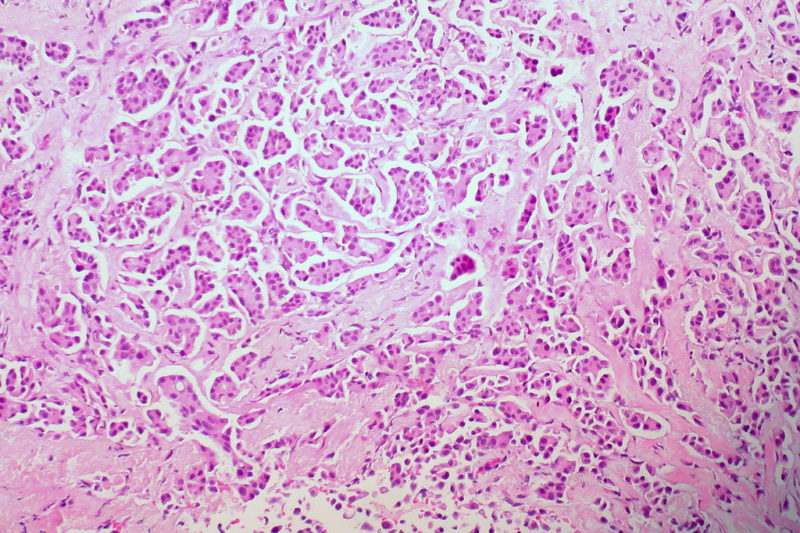
Cancers have long been classified by the part of the body they’re known to afflict – brain cancer, lung cancer, liver cancer, for example. But a new drug – Pembrolizumab – takes a different approach. Also called Keytruda, the drug targets cancer cells and reveals them to the immune system, wherever they are in the body.
The results of clinical trials have been very positive and New York Times Science and Medicine Reporter Gina Kolata says Keytruda has been approved for all types of cancers as long as they have certain mutations that spew out distorted proteins.
“If you have this type of mutation, then your immune system – given this drug – can start recognizing your cancer as something foreign and obliterate it,” Kolata says.
Kolata says the results of trials that included 86 patients were so “astounding” that the FDA promptly allowed Keytruda to be placed on the market. However, the drug works for just four percent of cancer patients.
What you’ll hear in this segment:
– Which patients qualify for this drug
– What type of genetic test is required for treatment
– How much the drug costs
– What are the next steps for this drug
Written by Louise Rodriguez.